
WELCOME
Chemistry
The scientific study of matter, its properties, how it behaves, and the changes it undergoes
Chemistry 10
Like I said that in the previous article, chemistry in Grade 10 in palmer is taught as part of the Science 10 course. Science 10 is a mandatory course for all students in grade 10. Students will learn atomic structure, the periodic table, chemical bonding, and the differences between ionic and covalent compounds. Students also learn how to name compounds, write chemical formulas, and balance chemical equations, and studying acids, bases, and common chemical reactions.
Ionic and covalent compounds
Ionic compounds form when a metal transfers electrons to a non‑metal. Because one atom loses electrons and the other gains them, they become charged particles called ions. This creates ionic compound. Examples include table salt (NaCl) and magnesium oxide (MgO).
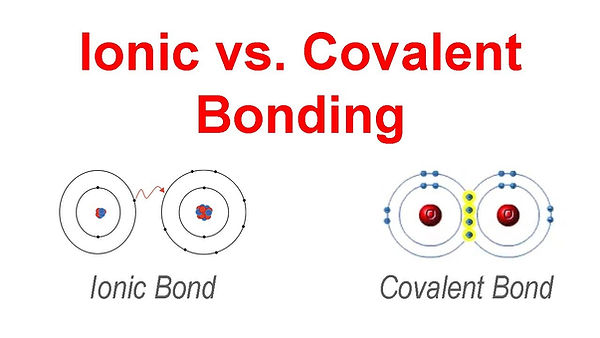
Covalent compounds, form when two non‑metals share electrons instead of transferring them. Examples include water (H₂O) and carbon dioxide (CO₂).
Balancing Chemical Equations
Balancing chemical equations is all about the number of each type of atom need to be the same on both sides of the equation. This follows the Law of Conservation of Mass, which says matter cannot be created or destroyed. When you balance an equation, you adjust the coefficients (the big numbers in front of compounds), not the subscripts (the small numbers inside formulas).
Here is a example of how to balance a chemical equation:

1. Write the unbalanced equation
Example:
H2+O2→H2O
2. Count atoms on both sides
-
Left: H = 2, O = 2
-
Right: H = 2, O = 1
3. Balance one element at a time
Oxygen is unbalanced, so put a 2 in front of H₂O:
H2+O2→2H2O
4. Balance hydrogen
Put a 2 in front of H₂:
2H2+O2→2H2O
Now both sides match.
5. Double‑check your work
-
Left: H = 4, O = 2
-
Right: H = 4, O = 2
Both side is now balanced.

.png)