
WELCOME
Chemistry
The scientific study of matter, its properties, how it behaves, and the changes it undergoes
Chemistry 8-9

Like I said that in the previous article, chemistry in Grades 8 to 10 in palmer is taught as part of the Science 8 and Science 9 course it is not a separate course. Students can learn foundational chemistry topics like the particle model of matter, states and changes of matter, elements and compounds, and basic atomic theory in Grade 8. In Grade 9, the chemistry mainly is about atoms and ions, chemical formulas, naming compounds, reaction types, and the periodic table(Last article)
Chemistry 8
The particle model of matter
The particle model of matter explains what all substances are made of and how they behave. It states that all matter is made of atoms that are moving around. The movement depends on the temperature, hotter the particles(atoms) the faster they move, lower the temperature slower they will move. There are some spaces between the particles(atoms) , and the amount of space changes depending on whether the substance is a solid, liquid, or gas. In solid, the particles have little energy, so they were vibrating. For liquid, the particles is starting to move much faster, and taking up more space than they were solid. Gas is when the particles move the fastest which also take p the most space. The model also says that how the particles attract each other, why solids hold their shape, why liquids can flow, and why gases spread out to fill any space.


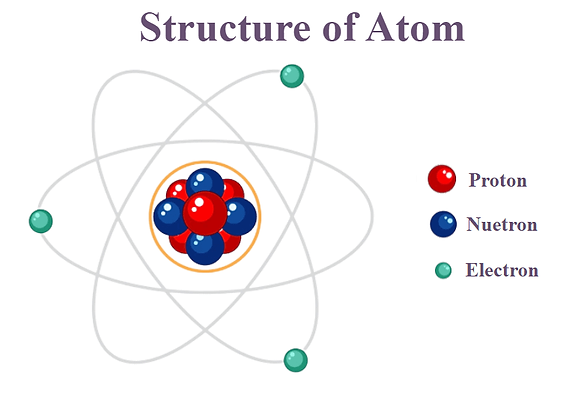
Basic Atomic Theory
Atomic theory is about that all matter is made of tiny particles called atoms, and each element is made of its own type of atom. Atoms can join together to form molecules and compounds, and they are the basic building blocks of everything around you.

Grade 9
Atoms and Ions
Atoms are the tiny particles that make up all things that is around you. An atom has a nucleus with protons and neutrons inside it, and electrons that move around it. Protons and neutrons took up most of the mass of a element and electron have almost no mass(More explanation in the last article) Atoms are neutral because they have the same number of protons and electrons.


Metals
Metals
However, some atoms are negatively charge (Mostly non-metals elements) and positively charge (mostly Metals elements) , they are call ions. If an atom loses electrons, it becomes a positive ion. If it gains electrons, it becomes a negative ion. Ion mostly form when 2 atoms try to combine into a compound, which make up most of the stuff that we used in our lives, for example, Nacl, which is table salt.